What Are Equivalent Hydrogen Atoms
 | |
| Full general | |
|---|---|
| Symbol | iH |
| Names | hydrogen atom, H-i, protium |
| Protons (Z) | ane |
| Neutrons (North) | 0 |
| Nuclide data | |
| Natural affluence | 99.985% |
| Half-life (t 1/2) | stable |
| Isotope mass | 1.007825 Da |
| Spin | i / 2 |
| Excess free energy | vii288.969±0.001 keV |
| Binding energy | 0.000±0.0000 keV |
| Isotopes of hydrogen Complete table of nuclides | |
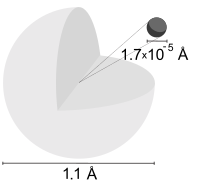
Depiction of a hydrogen atom showing the diameter as about twice the Bohr model radius. (Image not to scale)
A hydrogen atom is an atom of the chemical chemical element hydrogen. The electrically neutral cantlet contains a single positively charged proton and a single negatively charged electron spring to the nucleus past the Coulomb force. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.[i]
In everyday life on Earth, isolated hydrogen atoms (chosen "atomic hydrogen") are extremely rare. Instead, a hydrogen atom tends to combine with other atoms in compounds, or with another hydrogen atom to form ordinary (diatomic) hydrogen gas, H2. "Atomic hydrogen" and "hydrogen atom" in ordinary English use have overlapping, nevertheless singled-out, meanings. For example, a h2o molecule contains ii hydrogen atoms, but does non contain atomic hydrogen (which would refer to isolated hydrogen atoms).
Atomic spectroscopy shows that there is a detached infinite set of states in which a hydrogen (or any) cantlet can exist, contrary to the predictions of classical physics. Attempts to develop a theoretical understanding of the states of the hydrogen atom have been important to the history of quantum mechanics, since all other atoms can be roughly understood by knowing in detail most this simplest diminutive construction.
Isotopes [edit]
The nigh abundant isotope, hydrogen-1, protium, or light hydrogen, contains no neutrons and is but a proton and an electron. Protium is stable and makes up 99.985% of naturally occurring hydrogen atoms.[2]
Deuterium contains one neutron and one proton in its nucleus. Deuterium is stable and makes upwardly 0.0156% of naturally occurring hydrogen[2] and is used in industrial processes similar nuclear reactors and Nuclear Magnetic Resonance.
Tritium contains two neutrons and one proton in its nucleus and is not stable, decomposable with a one-half-life of 12.32 years. Because of its short half-life, tritium does not exist in nature except in trace amounts.
Heavier isotopes of hydrogen are simply created artificially in particle accelerators and have half-lives on the club of 10−22 seconds. They are unbound resonances located across the neutron drip line; this results in prompt emission of a neutron.
The formulas below are valid for all three isotopes of hydrogen, merely slightly different values of the Rydberg constant (correction formula given below) must be used for each hydrogen isotope.
Hydrogen ion [edit]
Lone neutral hydrogen atoms are rare nether normal conditions. However, neutral hydrogen is common when it is covalently bound to another atom, and hydrogen atoms can also exist in cationic and anionic forms.
If a neutral hydrogen atom loses its electron, it becomes a cation. The resulting ion, which consists solely of a proton for the usual isotope, is written as "H+" and sometimes called hydron. Free protons are common in the interstellar medium, and solar air current. In the context of aqueous solutions of classical Brønsted–Lowry acids, such as muriatic acid, it is actually hydronium, H3O+, that is meant. Instead of a literal ionized unmarried hydrogen atom being formed, the acrid transfers the hydrogen to H2O, forming HthreeO+.
If instead a hydrogen atom gains a second electron, it becomes an anion. The hydrogen anion is written as "H–" and called hydride.
Theoretical analysis [edit]
The hydrogen atom has special significance in quantum mechanics and quantum field theory equally a simple 2-body problem physical system which has yielded many simple analytical solutions in closed-class.
Failed classical description [edit]
Experiments by Ernest Rutherford in 1909 showed the structure of the atom to be a dumbo, positive nucleus with a tenuous negative charge cloud effectually information technology. This immediately raised questions about how such a system could be stable. Classical electromagnetism had shown that any accelerating accuse radiates free energy, every bit shown by the Larmor formula. If the electron is assumed to orbit in a perfect circumvolve and radiates energy continuously, the electron would rapidly spiral into the nucleus with a fall time of:[3]
where is the Bohr radius and is the classical electron radius. If this were truthful, all atoms would instantly collapse, however atoms seem to be stable. Furthermore, the spiral inward would release a smear of electromagnetic frequencies as the orbit got smaller. Instead, atoms were observed to only emit discrete frequencies of radiation. The resolution would lie in the development of quantum mechanics.
Bohr–Sommerfeld Model [edit]
In 1913, Niels Bohr obtained the energy levels and spectral frequencies of the hydrogen atom later making a number of unproblematic assumptions in order to correct the failed classical model. The assumptions included:
- Electrons can only exist in certain, detached circular orbits or stationary states, thereby having a discrete set of possible radii and energies.
- Electrons practise not emit radiation while in ane of these stationary states.
- An electron tin can gain or lose free energy by jumping from one discrete orbit to another.
Bohr supposed that the electron'south athwart momentum is quantized with possible values:
where and is Planck abiding over . He besides supposed that the centripetal force which keeps the electron in its orbit is provided past the Coulomb force, and that energy is conserved. Bohr derived the energy of each orbit of the hydrogen atom to be:[iv]
where is the electron mass, is the electron charge, is the vacuum permittivity, and is the breakthrough number (now known as the chief quantum number). Bohr's predictions matched experiments measuring the hydrogen spectral series to the first society, giving more conviction to a theory that used quantized values.
For , the value[5]
is called the Rydberg unit of free energy. Information technology is related to the Rydberg abiding of atomic physics past
The verbal value of the Rydberg abiding assumes that the nucleus is infinitely massive with respect to the electron. For hydrogen-1, hydrogen-2 (deuterium), and hydrogen-3 (tritium) which take finite mass, the abiding must exist slightly modified to use the reduced mass of the system, rather than but the mass of the electron. This includes the kinetic energy of the nucleus in the trouble, considering the total (electron plus nuclear) kinetic energy is equivalent to the kinetic free energy of the reduced mass moving with a velocity equal to the electron velocity relative to the nucleus. However, since the nucleus is much heavier than the electron, the electron mass and reduced mass are nearly the same. The Rydberg abiding RGrand for a hydrogen cantlet (one electron), R is given by
where is the mass of the atomic nucleus. For hydrogen-ane, the quantity is well-nigh i/1836 (i.due east. the electron-to-proton mass ratio). For deuterium and tritium, the ratios are nigh ane/3670 and 1/5497 respectively. These figures, when added to i in the denominator, correspond very small corrections in the value of R, and thus only small corrections to all free energy levels in corresponding hydrogen isotopes.
There were yet bug with Bohr's model:
- it failed to predict other spectral details such as fine structure and hyperfine structure
- information technology could merely predict energy levels with any accuracy for unmarried–electron atoms (hydrogen-like atoms)
- the predicted values were only correct to , where is the fine-structure constant.
Nearly of these shortcomings were resolved by Arnold Sommerfeld'due south modification of the Bohr model. Sommerfeld introduced two boosted degrees of freedom, allowing an electron to motility on an elliptical orbit characterized by its eccentricity and declination with respect to a chosen centrality. This introduced two boosted quantum numbers, which stand for to the orbital angular momentum and its projection on the called axis. Thus the correct multiplicity of states (except for the factor 2 accounting for the yet unknown electron spin) was found. Further, by applying special relativity to the elliptic orbits, Sommerfeld succeeded in deriving the correct expression for the fine structure of hydrogen spectra (which happens to exist exactly the same as in the most elaborate Dirac theory). However, some observed phenomena, such as the anomalous Zeeman effect, remained unexplained. These issues were resolved with the total development of quantum mechanics and the Dirac equation. It is often alleged that the Schrödinger equation is superior to the Bohr–Sommerfeld theory in describing hydrogen atom. This is not the example, as well-nigh of the results of both approaches coincide or are very close (a remarkable exception is the trouble of hydrogen atom in crossed electrical and magnetic fields, which cannot be self-consistently solved in the framework of the Bohr–Sommerfeld theory), and in both theories the master shortcomings event from the absenteeism of the electron spin. It was the complete failure of the Bohr–Sommerfeld theory to explain many-electron systems (such as helium atom or hydrogen molecule) which demonstrated its inadequacy in describing breakthrough phenomena.
Schrödinger equation [edit]
The Schrödinger equation allows i to calculate the stationary states and besides the time development of quantum systems. Exact analytical answers are available for the nonrelativistic hydrogen atom. Earlier we get to present a formal business relationship, here we give an elementary overview.
Given that the hydrogen atom contains a nucleus and an electron, quantum mechanics allows ane to predict the probability of finding the electron at whatsoever given radial altitude . It is given past the square of a mathematical function known as the "wavefunction," which is a solution of the Schrödinger equation. The lowest energy equilibrium country of the hydrogen cantlet is known equally the ground country. The footing state wave office is known every bit the wavefunction. It is written as:
Here, is the numerical value of the Bohr radius. The probability density of finding the electron at a altitude in any radial direction is the squared value of the wavefunction:
The wavefunction is spherically symmetric, and the expanse of a crush at distance is , so the total probability of the electron beingness in a shell at a distance and thickness is
It turns out that this is a maximum at . That is, the Bohr picture of an electron orbiting the nucleus at radius corresponds to the well-nigh probable radius. Actually, in that location is a finite probability that the electron may be found at whatever identify , with the probability indicated by the foursquare of the wavefunction. Since the probability of finding the electron somewhere in the whole volume is unity, the integral of is unity. Then we say that the wavefunction is properly normalized.
Every bit discussed below, the ground state is likewise indicated by the quantum numbers . The second lowest energy states, just above the ground state, are given by the quantum numbers , , and . These states all take the aforementioned energy and are known as the and states. There is i state:
and in that location are three states:
An electron in the or land is almost likely to be establish in the second Bohr orbit with free energy given by the Bohr formula.
Wavefunction [edit]
The Hamiltonian of the hydrogen atom is the radial kinetic energy operator and Coulomb attraction force between the positive proton and negative electron. Using the time-independent Schrödinger equation, ignoring all spin-coupling interactions and using the reduced mass , the equation is written as:
Expanding the Laplacian in spherical coordinates:
This is a separable, fractional differential equation which can be solved in terms of special functions. When the wavefunction is separated as product of functions , , and three independent differential functions appears[vi] with A and B being the separation constants:
- radial:
- polar:
- azimuth:
The normalized position wavefunctions, given in spherical coordinates are:
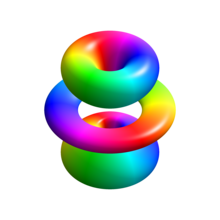
3D analogy of the eigenstate . Electrons in this state are 45% probable to exist found inside the solid body shown.
where:
The quantum numbers can take the following values:
- (principal breakthrough number)
- (azimuthal quantum number)
- (magnetic quantum number).
Additionally, these wavefunctions are normalized (i.e., the integral of their modulus square equals ane) and orthogonal:
where is the country represented by the wavefunction in Dirac annotation, and is the Kronecker delta part.[xi]
The wavefunctions in momentum space are related to the wavefunctions in position infinite through a Fourier transform
which, for the jump states, results in[12]
where denotes a Gegenbauer polynomial and is in units of .
The solutions to the Schrödinger equation for hydrogen are analytical, giving a elementary expression for the hydrogen energy levels and thus the frequencies of the hydrogen spectral lines and fully reproduced the Bohr model and went beyond it. It also yields two other quantum numbers and the shape of the electron's wave function ("orbital") for the various possible quantum-mechanical states, thus explaining the anisotropic character of atomic bonds.
The Schrödinger equation also applies to more than complicated atoms and molecules. When there is more one electron or nucleus the solution is not belittling and either estimator calculations are necessary or simplifying assumptions must be made.
Since the Schrödinger equation is but valid for not-relativistic quantum mechanics, the solutions it yields for the hydrogen atom are not entirely right. The Dirac equation of relativistic quantum theory improves these solutions (see below).
Results of Schrödinger equation [edit]
The solution of the Schrödinger equation (moving ridge equation) for the hydrogen atom uses the fact that the Coulomb potential produced by the nucleus is isotropic (it is radially symmetric in space and only depends on the distance to the nucleus). Although the resulting energy eigenfunctions (the orbitals) are non necessarily isotropic themselves, their dependence on the athwart coordinates follows completely generally from this isotropy of the underlying potential: the eigenstates of the Hamiltonian (that is, the energy eigenstates) can exist chosen as simultaneous eigenstates of the angular momentum operator. This corresponds to the fact that angular momentum is conserved in the orbital motion of the electron effectually the nucleus. Therefore, the energy eigenstates may be classified by ii angular momentum quantum numbers, and (both are integers). The angular momentum quantum number determines the magnitude of the angular momentum. The magnetic quantum number determines the projection of the angular momentum on the (arbitrarily chosen) -centrality.
In add-on to mathematical expressions for full angular momentum and angular momentum projection of wavefunctions, an expression for the radial dependence of the moving ridge functions must be found. It is only here that the details of the Coulomb potential enter (leading to Laguerre polynomials in ). This leads to a third quantum number, the principal quantum number . The principal quantum number in hydrogen is related to the atom'south total free energy.
Note that the maximum value of the athwart momentum breakthrough number is express by the principal breakthrough number: it can run only up to , i.e., .
Due to angular momentum conservation, states of the same but different have the same energy (this holds for all issues with rotational symmetry). In addition, for the hydrogen atom, states of the same only different are also degenerate (i.eastward., they have the same energy). Nonetheless, this is a specific property of hydrogen and is no longer truthful for more complicated atoms which have an (effective) potential differing from the form (due to the presence of the inner electrons shielding the nucleus potential).
Taking into account the spin of the electron adds a final quantum number, the projection of the electron's spin angular momentum forth the -axis, which tin accept on two values. Therefore, any eigenstate of the electron in the hydrogen cantlet is described fully by four quantum numbers. Co-ordinate to the usual rules of quantum mechanics, the actual state of the electron may be whatsoever superposition of these states. This explains likewise why the choice of -axis for the directional quantization of the angular momentum vector is immaterial: an orbital of given and obtained for another preferred axis can ever exist represented as a suitable superposition of the various states of unlike (but aforementioned ) that have been obtained for .
Mathematical summary of eigenstates of hydrogen atom [edit]
In 1928, Paul Dirac plant an equation that was fully compatible with special relativity, and (as a event) made the moving ridge function a 4-component "Dirac spinor" including "upward" and "downwardly" spin components, with both positive and "negative" energy (or affair and antimatter). The solution to this equation gave the following results, more accurate than the Schrödinger solution.
Energy levels [edit]
The free energy levels of hydrogen, including fine construction (excluding Lamb shift and hyperfine structure), are given by the Sommerfeld fine-structure expression:[xiii]
where is the fine-structure constant and is the full angular momentum quantum number, which is equal to , depending on the orientation of the electron spin relative to the orbital angular momentum.[fourteen] This formula represents a small correction to the free energy obtained by Bohr and Schrödinger every bit given above. The gene in square brackets in the terminal expression is nigh 1; the actress term arises from relativistic effects (for details, see #Features going beyond the Schrödinger solution). It is worth noting that this expression was beginning obtained past A. Sommerfeld in 1916 based on the relativistic version of the erstwhile Bohr theory. Sommerfeld has however used different notation for the quantum numbers.
Coherent states [edit]
The coherent states have been proposed as[15]
which satisfies and takes the form
Visualizing the hydrogen electron orbitals [edit]
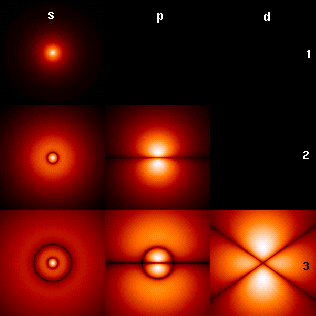
Probability densities through the xz-plane for the electron at different quantum numbers (ℓ, across top; north, downwardly side; m = 0)
The epitome to the right shows the start few hydrogen atom orbitals (free energy eigenfunctions). These are cross-sections of the probability density that are colour-coded (black represents zero density and white represents the highest density). The angular momentum (orbital) quantum number ℓ is denoted in each column, using the usual spectroscopic alphabetic character code (s means ℓ = 0, p means ℓ = 1, d means ℓ = ii). The main (main) quantum number n (= 1, 2, 3, ...) is marked to the right of each row. For all pictures the magnetic quantum number m has been set to 0, and the cross-sectional plane is the xz-airplane (z is the vertical axis). The probability density in three-dimensional space is obtained past rotating the one shown here around the z-axis.
The "footing country", i.due east. the state of lowest energy, in which the electron is usually plant, is the first one, the 1s state (principal quantum level north = i, ℓ = 0).
Black lines occur in each only the first orbital: these are the nodes of the wavefunction, i.e. where the probability density is zero. (More than precisely, the nodes are spherical harmonics that appear as a result of solving the Schrödinger equation in spherical coordinates.)
The quantum numbers determine the layout of these nodes.[16] There are:
Features going beyond the Schrödinger solution [edit]
In that location are several important effects that are neglected by the Schrödinger equation and which are responsible for sure pocket-size but measurable deviations of the existent spectral lines from the predicted ones:
- Although the mean speed of the electron in hydrogen is only 1/137th of the speed of light, many modern experiments are sufficiently precise that a complete theoretical explanation requires a fully relativistic treatment of the problem. A relativistic treatment results in a momentum increase of most 1 part in 37,000 for the electron. Since the electron's wavelength is determined by its momentum, orbitals containing college speed electrons prove contraction due to smaller wavelengths.
- Even when at that place is no external magnetic field, in the inertial frame of the moving electron, the electromagnetic field of the nucleus has a magnetic component. The spin of the electron has an associated magnetic moment which interacts with this magnetic field. This consequence is also explained by special relativity, and it leads to the so-called spin–orbit coupling, i.e., an interaction between the electron'southward orbital move around the nucleus, and its spin.
Both of these features (and more) are incorporated in the relativistic Dirac equation, with predictions that come up still closer to experiment. Again the Dirac equation may exist solved analytically in the special case of a two-body system, such as the hydrogen atom. The resulting solution quantum states now must exist classified by the total angular momentum number j (arising through the coupling between electron spin and orbital athwart momentum). States of the same j and the same n are nevertheless degenerate. Thus, direct analytical solution of Dirac equation predicts 2S( 1 / ii ) and 2P( 1 / 2 ) levels of hydrogen to accept exactly the same energy, which is in a contradiction with observations (Lamb–Retherford experiment).
- In that location are always vacuum fluctuations of the electromagnetic field, according to quantum mechanics. Due to such fluctuations degeneracy between states of the aforementioned j only dissimilar fifty is lifted, giving them slightly different energies. This has been demonstrated in the famous Lamb–Retherford experiment and was the starting point for the development of the theory of breakthrough electrodynamics (which is able to deal with these vacuum fluctuations and employs the famous Feynman diagrams for approximations using perturbation theory). This effect is now chosen Lamb shift.
For these developments, it was essential that the solution of the Dirac equation for the hydrogen atom could be worked out exactly, such that any experimentally observed divergence had to exist taken seriously as a signal of failure of the theory.
Alternatives to the Schrödinger theory [edit]
In the language of Heisenberg's matrix mechanics, the hydrogen atom was first solved by Wolfgang Pauli[17] using a rotational symmetry in four dimensions [O(4)-symmetry] generated by the angular momentum and the Laplace–Runge–Lenz vector. Past extending the symmetry group O(4) to the dynamical group O(4,two), the unabridged spectrum and all transitions were embedded in a single irreducible group representation.[18]
In 1979 the (non-relativistic) hydrogen cantlet was solved for the get-go time within Feynman's path integral conception of breakthrough mechanics by Duru and Kleinert.[19] [20] This piece of work greatly extended the range of applicability of Feynman'southward method.
Encounter also [edit]
- Antihydrogen
- Atomic orbital
- Balmer series
- Helium cantlet
- Lithium cantlet
- Hydrogen molecular ion
- Proton decay
- Quantum chemistry
- Quantum state
- Theoretical and experimental justification for the Schrödinger equation
- Trihydrogen cation
- List of quantum-mechanical systems with belittling solutions
References [edit]
- ^ Palmer, D. (thirteen September 1997). "Hydrogen in the Universe". NASA. Archived from the original on 29 October 2014. Retrieved 23 February 2017.
- ^ a b Housecroft, Catherine E.; Sharpe, Alan G. (2005). Inorganic Chemistry (second ed.). Pearson Prentice-Hall. p. 237. ISBN0130-39913-ii.
- ^ Olsen, James; McDonald, Kirk (7 March 2005). "Classical Lifetime of a Bohr Atom" (PDF). Joseph Henry Laboratories, Princeton Academy.
- ^ "Derivation of Bohr's Equations for the Ane-electron Atom" (PDF). University of Massachusetts Boston.
- ^ Eite Tiesinga, Peter J. Mohr, David B. Newell, and Barry N. Taylor (2019), "The 2018 CODATA Recommended Values of the Fundamental Concrete Constants" (Spider web Version 8.0). Database developed by J. Baker, M. Douma, and South. Kotochigova. Available at http://physics.nist.gov/constants, National Establish of Standards and Technology, Gaithersburg, Doctor 20899. Link to R∞, Link to hcR∞
- ^ "Solving Schrödinger'due south equation for the hydrogen atom :: Atomic Physics :: Rudi Winter's web space". users.aber.ac.uk . Retrieved 30 November 2020.
- ^ Messiah, Albert (1999). Breakthrough Mechanics. New York: Dover. p. 1136. ISBN0-486-40924-4.
- ^ LaguerreL. Wolfram Mathematica folio
- ^ Griffiths, p. 152
- ^ Condon and Shortley (1963). The Theory of Atomic Spectra. London: Cambridge. p. 441.
- ^ Griffiths, Ch. 4 p. 89
- ^ Bransden, B. H.; Joachain, C. J. (1983). Physics of Atoms and Molecules. Longman. p. Appendix 5. ISBN0-582-44401-two.
- ^ Sommerfeld, Arnold (1919). Atombau und Spektrallinien [Atomic Construction and Spectral Lines]. Braunschweig: Friedrich Vieweg und Sohn. ISBNthree-87144-484-7. German English
- ^ Atkins, Peter; de Paula, Julio (2006). Physical Chemistry (8th ed.). W. H. Freeman. p. 349. ISBN0-7167-8759-viii.
- ^ Klauder, John R (21 June 1996). "Coherent states for the hydrogen atom". Journal of Physics A: Mathematical and General. 29 (12): L293–L298. arXiv:quant-ph/9511033. doi:x.1088/0305-4470/29/12/002. S2CID 14124660.
- ^ Summary of diminutive breakthrough numbers. Lecture notes. 28 July 2006
- ^ Pauli, Due west (1926). "Über das Wasserstoffspektrum vom Standpunkt der neuen Quantenmechanik". Zeitschrift für Physik. 36 (five): 336–363. Bibcode:1926ZPhy...36..336P. doi:x.1007/BF01450175. S2CID 128132824.
- ^ Kleinert H. (1968). "Grouping Dynamics of the Hydrogen Atom" (PDF). Lectures in Theoretical Physics, Edited by W.Due east. Brittin and A.O. Barut, Gordon and Breach, N.Y. 1968: 427–482.
- ^ Duru I.H., Kleinert H. (1979). "Solution of the path integral for the H-atom" (PDF). Physics Messages B. 84 (2): 185–188. Bibcode:1979PhLB...84..185D. doi:x.1016/0370-2693(79)90280-6.
- ^ Duru I.H., Kleinert H. (1982). "Breakthrough Mechanics of H-Atom from Path Integrals" (PDF). Fortschr. Phys. 30 (2): 401–435. Bibcode:1982ForPh..thirty..401D. doi:10.1002/prop.19820300802.
Books [edit]
- Griffiths, David J. (1995). Introduction to Quantum Mechanics. Prentice Hall. ISBN0-xiii-111892-7. Department iv.2 deals with the hydrogen atom specifically, merely all of Chapter 4 is relevant.
- Kleinert, H. (2009). Path Integrals in Quantum Mechanics, Statistics, Polymer Physics, and Fiscal Markets, quaternary edition, Worldscibooks.com, Globe Scientific, Singapore (as well available online physik.fu-berlin.de)
External links [edit]
- Physics of hydrogen atom on Scienceworld
What Are Equivalent Hydrogen Atoms,
Source: https://en.wikipedia.org/wiki/Hydrogen_atom
Posted by: parishmanthaten.blogspot.com











































![{\displaystyle -{\frac {\hbar ^{2}}{2\mu }}\left[{\frac {1}{r^{2}}}{\frac {\partial }{\partial r}}\left(r^{2}{\frac {\partial \psi }{\partial r}}\right)+{\frac {1}{r^{2}\sin \theta }}{\frac {\partial }{\partial \theta }}\left(\sin \theta {\frac {\partial \psi }{\partial \theta }}\right)+{\frac {1}{r^{2}\sin ^{2}\theta }}{\frac {\partial ^{2}\psi }{\partial \varphi ^{2}}}\right]-{\frac {e^{2}}{4\pi \varepsilon _{0}r}}\psi =E\psi }](https://wikimedia.org/api/rest_v1/media/math/render/svg/fed150abb1693ab2493937b669446a54865b9562)





























![{\displaystyle {\begin{aligned}E_{j\,n}={}&-\mu c^{2}\left[1-\left(1+\left[{\frac {\alpha }{n-j-{\frac {1}{2}}+{\sqrt {\left(j+{\frac {1}{2}}\right)^{2}-\alpha ^{2}}}}}\right]^{2}\right)^{-1/2}\right]\\\approx {}&-{\frac {\mu c^{2}\alpha ^{2}}{2n^{2}}}\left[1+{\frac {\alpha ^{2}}{n^{2}}}\left({\frac {n}{j+{\frac {1}{2}}}}-{\frac {3}{4}}\right)\right],\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/9e54f0064eafaeab9e7d8b3e5e41e667a3138a7b)




![{\displaystyle {\begin{aligned}\langle r,\theta ,\varphi \mid s,\gamma ,{\bar {\Omega }}\rangle ={}&e^{-s^{2}/2}\sum _{n=0}^{\infty }(s^{n}e^{i\gamma /(n+1)^{2}}/{\sqrt {n!}})\\&{}\times \,\sum _{\ell =0}^{n}u_{n+1}^{\ell }(r)\sum _{m=-\ell }^{\ell }\left[{\frac {(2\ell )!}{(\ell +m)!(\ell -m)!}}\right]^{1/2}\left(\sin {\frac {\bar {\theta }}{2}}\right)^{\ell -m}\left(\cos {\frac {\bar {\theta }}{2}}\right)^{\ell +m}\\&{}\times \,e^{-i(m{\bar {\varphi }}+\ell {\bar {\psi }})}Y_{\ell m}(\theta ,\varphi ){\sqrt {2\ell +1}}.\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/bb48ff266b61e92b9bdfcd39d562729c3910e97a)

0 Response to "What Are Equivalent Hydrogen Atoms"
Post a Comment